Density and Thermal Expansivity of Molten 2LiF-BeF2 (FLiBe): Measurements and Uncertainty Quantification | Journal of Chemical & Engineering Data

SOLVED: Liquid N2 has a density of 875.4 kg m-3 at its normal boiling point. What volume does a balloon occupy at 298 K and a pressure of 1.00 atm if 5.25

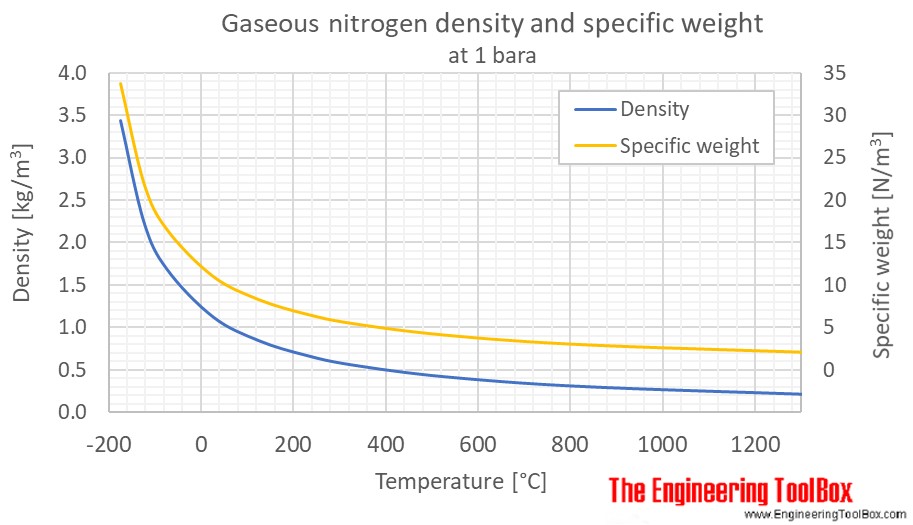
Liquid Nitrogen JerryHancock. What are the Risks? Density of Gases at STP (Standard Temperature Pressure): Nitrogen (N): 1.25 kg/m 3 Carbon Dioxide (CO. - ppt download

The density of liquid nitrogen is 0.807gmL−3. If a person acidently swallowed a .028mL drop of liquid nitrogen. What volume of nitrogen gas would be evolved in their body 100.0kPa and 27oC?
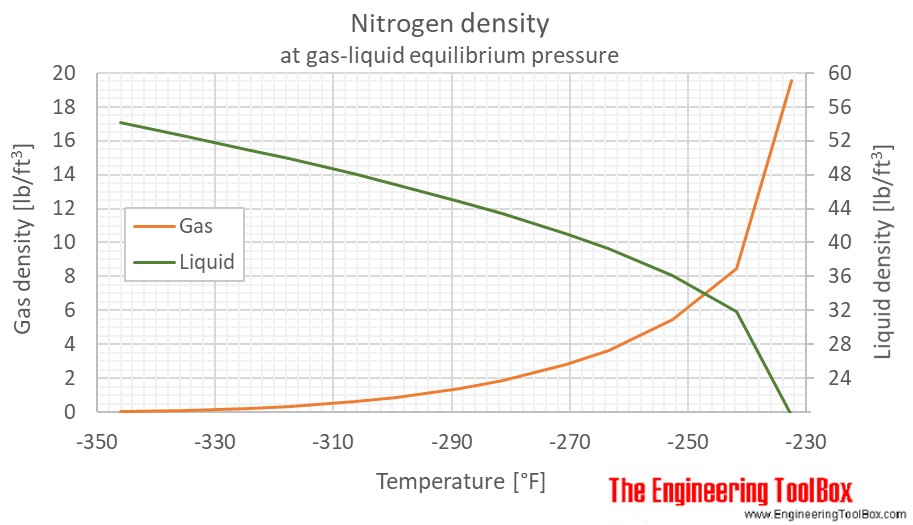

![Marathi] Calculate R.M.S speed of nitrogen at 0^@C. (Density of nitro Marathi] Calculate R.M.S speed of nitrogen at 0^@C. (Density of nitro](https://d10lpgp6xz60nq.cloudfront.net/ss/web-overlay-thumb/8557435.webp)


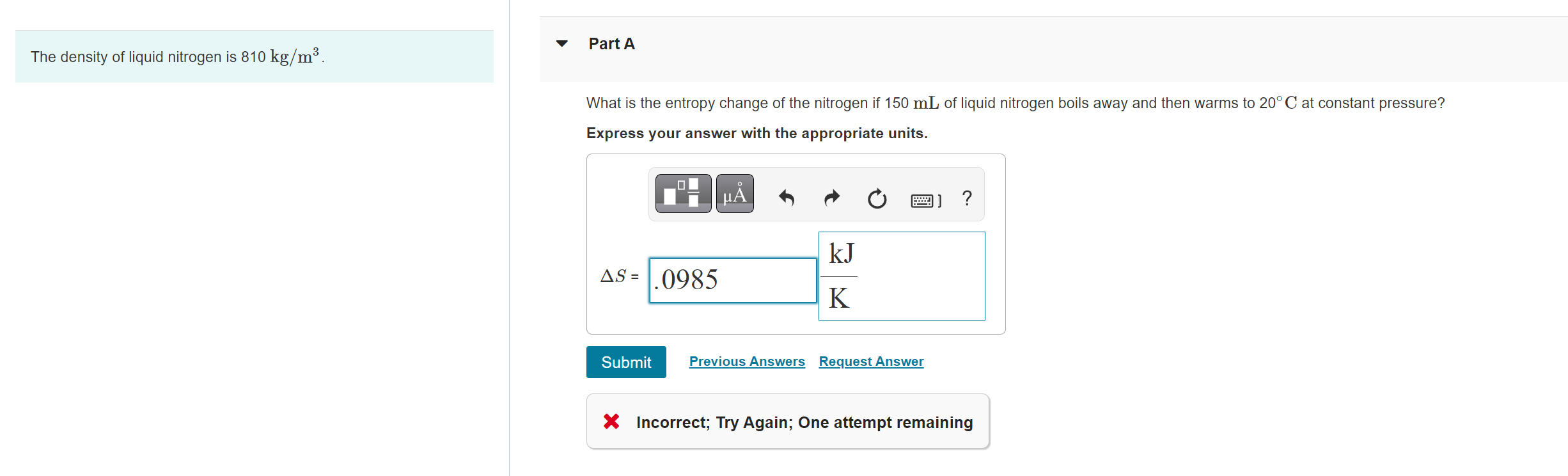




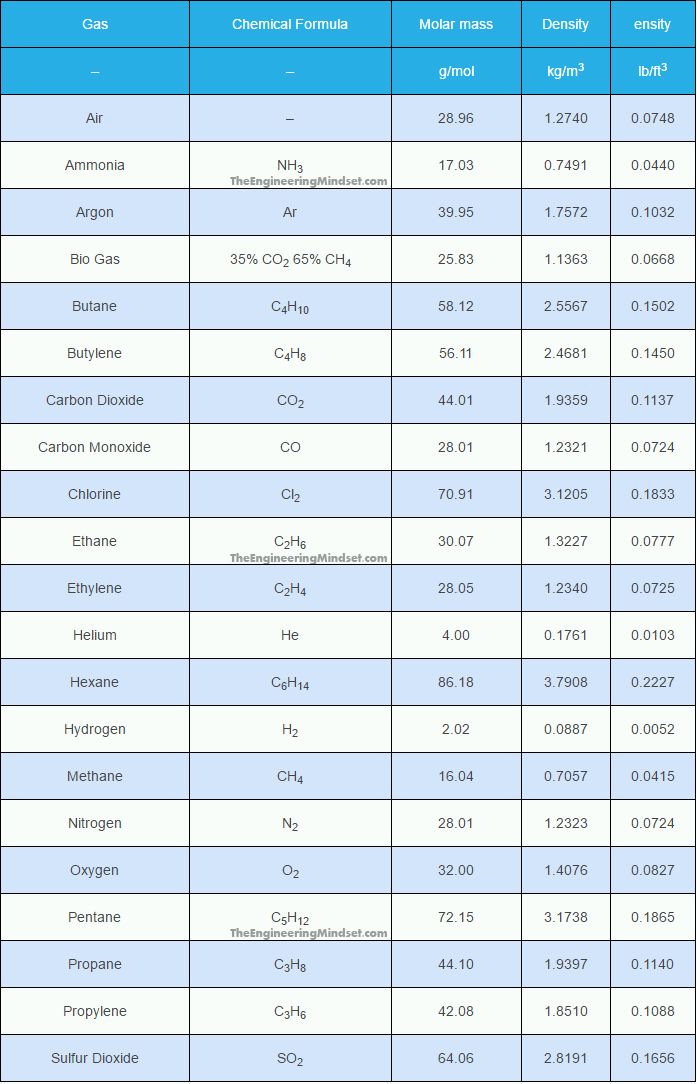
![Gas density variation [kg/m 3 ] vs. gas pressure and hydrogen... | Download Scientific Diagram Gas density variation [kg/m 3 ] vs. gas pressure and hydrogen... | Download Scientific Diagram](https://www.researchgate.net/publication/309226014/figure/fig3/AS:418676688867329@1476831709115/Gas-density-variation-kg-m-3-vs-gas-pressure-and-hydrogen-percentage-in-the-blend.png)