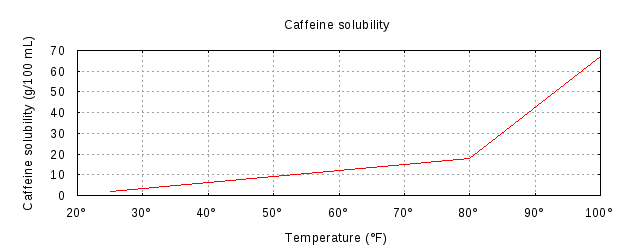
SOLVED: The solubility of caffeine in water at 100°C is equal to Circle the right answer Select one: Insoluble 18.0g/100ml 6.7g/100ml 2.2g/100ml

EP0158381B1 - A process for removing chlorogenic acid compounds and caffeine from coffee, and roasted coffee obtainable by this process - Google Patents
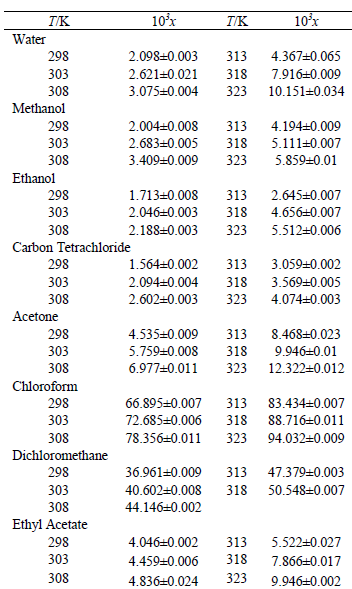
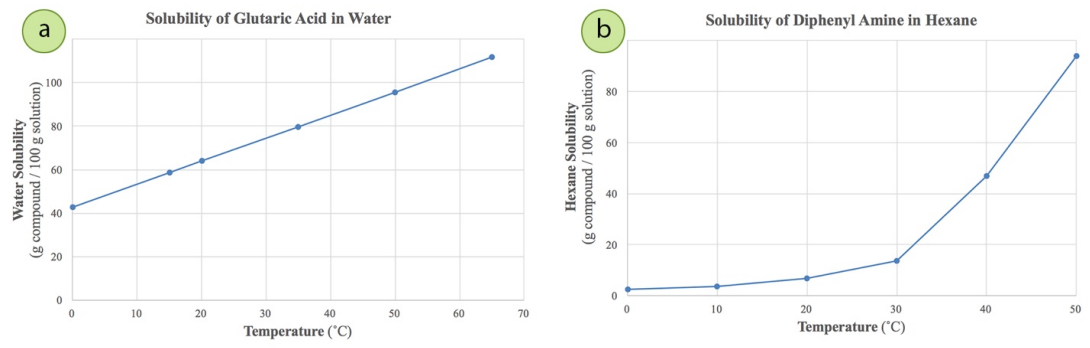
Solubility of caffeine in water, ethyl acetate, ethanol, carbon tetrachloride, methanol, chloroform, dichloromethane, and acetone between 298 and 323 K
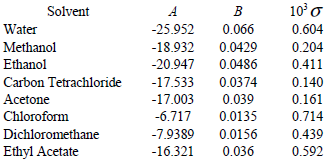
Solubility of caffeine in water, ethyl acetate, ethanol, carbon tetrachloride, methanol, chloroform, dichloromethane, and acetone between 298 and 323 K
![PDF] Demonstration of the Formation of the Caffeine-Dichloromethane-water Emulsion using Quantum Chemistry | Semantic Scholar PDF] Demonstration of the Formation of the Caffeine-Dichloromethane-water Emulsion using Quantum Chemistry | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/bbac6065c3ebf72dfa601dc108d61d0f0a7652b8/4-Table2-1.png)
PDF] Demonstration of the Formation of the Caffeine-Dichloromethane-water Emulsion using Quantum Chemistry | Semantic Scholar

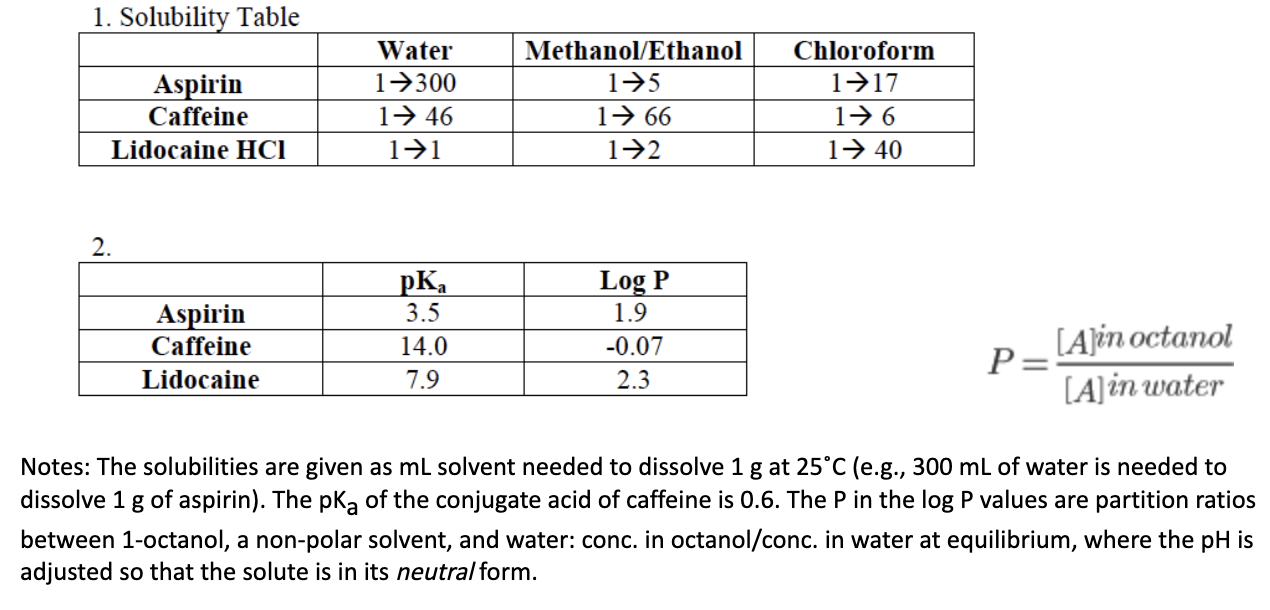
Caffeine (C8H10N4O2) is a stimulant found in coffees and teas. When dissolved in water, it can accept a - brainly.com
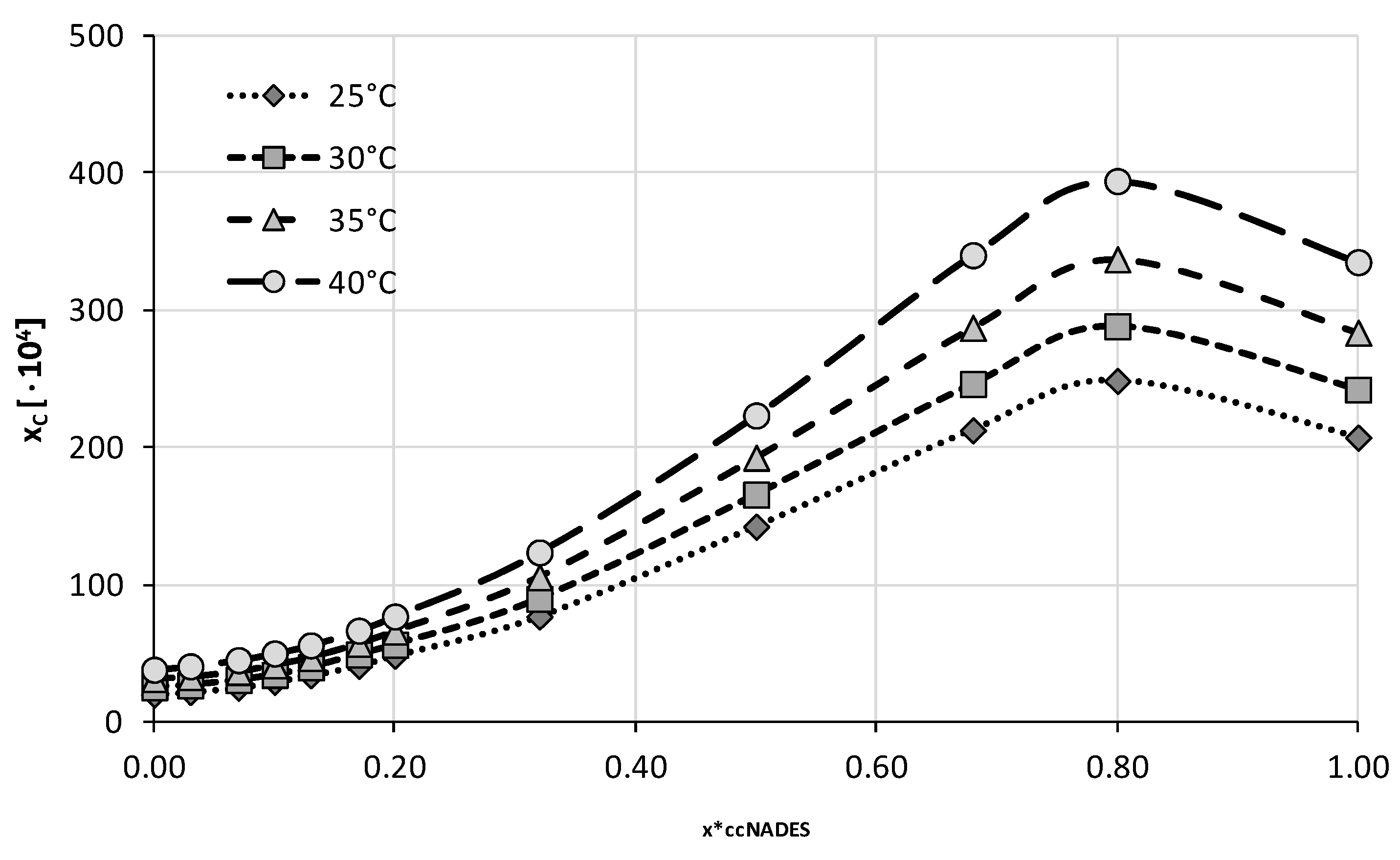
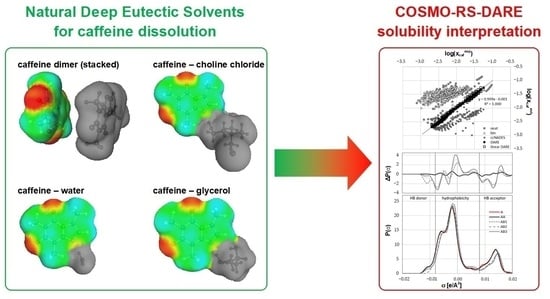
IJMS | Free Full-Text | Quantification of Caffeine Interactions in Choline Chloride Natural Deep Eutectic Solvents: Solubility Measurements and COSMO-RS-DARE Interpretation

0.070 g of caffeine is dissolved in 4.0 mL of water. The caffeine is extracted from the aqueous - YouTube

Table 2 from Solubility of Bioactive Substances in Ethyl Lactate + Water Mixtures: Ferulic Acid and Caffeine | Semantic Scholar

Figura 1-Caffeine solubility in water or water:IL mixtures (99.8:0.2,... | Download Scientific Diagram

When The Temperature Of The Water Is Increased The Amount Of Caffeine That Can Be Dissolved In It Also Increases

Solubility of caffeine in water, ethyl acetate, ethanol, carbon tetrachloride, methanol, chloroform, dichloromethane, and acetone between 298 and 323 K